News + Events Archive
2023

Tyler Godat Thesis Defense on January 26, 2023
9:00 a.m., 310 Gavett Hall
Functional Imaging and Classification of Retinal Ganglion Cells in the Living Primate Eye
Advisor: David Williams
The primate fovea is perhaps the most important location for vision in the retina, being specialized for high resolution spatial and chromatic vision, but it has been traditionally difficult to study in ex vivo electrophysiology and especially in vivo. The crucial visual properties conferred by the fovea are mediated by neurons called retinal ganglion cells, which exist in twenty or more distinct types that all tile the retina and are present from the far periphery up to the inner edge of the foveal slope. Microelectrode recordings of foveal retinal ganglion cells have been challenging due to their reduced size and the fragility of tissue survival in excised retina. Some of these difficulties in observing individual cells have been overcome in the in vivo retina by combining high resolution fluorescence adaptive optics ophthalmoscopy with calcium imaging to optically record functional responses in the living primate eye. The work in this thesis attempts to advance knowledge of functional properties of retinal ganglion cells at the primate fovea by studying their responses to various stimuli in the living primate (macaca fascicularis) eye. This includes measurements of retinal ganglion cells’ chromatic tuning and cone weightings, made possible by silent substitution style uniform chromatic flicker stimuli, and measurements of cells’ spatial transfer functions, made possible by monochromatic drifting gratings of various spatial frequencies. In particular, the most common type of retinal ganglion cell, the midget, is studied extensively to generate a broader understanding of the specialization and importance of the primate fovea. This work thus provides a closer look at the in situ spatial, chromatic, and temporal properties of these retinal ganglion cells than ever before possible. A novel stimulation delivery arm as part of a dual adaptive optics ophthalmoscope is also developed as part of this work to improve the resolution and breadth of stimuli used to interrogate the functional properties of retinal ganglion cells. These imaging and stimulation techniques provide a suite of tools for assessing and characterizing the functional properties of retinal ganglion cells in vivo which will inform the next generation of vision restoration strategies such as optogenetics and retinal prostheses.
2022

Jesse Schallek announced as new Director of ARIA
We are excited to announce that Jesse Schallek is the new Director of ARIA. Jesse's combination of scientific insight, engineering advances, interpersonal relationships and creative drive makes him an ideal fit to lead ARIA to new and exciting discoveries. We thank Jennifer Hunter for her 11 years of leadership and wish her well as she embarks on new research directions at the University of Waterloo.

2021

Williams inducted into National Academy of Inventors
The National Academy of Inventors is a member organization comprising US and international universities, and governmental and non-profit research institutes. It was founded in 2010 to recognize and encourage inventors with patents issued from the US Patent and Trademark Office, enhance the visibility of academic technology and innovation, encourage the disclosure of intellectual property, educate and mentor innovative students, and translate the inventions of its members to benefit society.
Previous NAI fellows from the University of Rochester are Kevin Parker, the William F. May Professor and dean emeritus of engineering and applied sciences; Wayne Knox, professor of optics; Jim Zavislan, professor of optics, and Jannick Rolland, the Brian J. Thompson Professor of Optical Engineering.
The most efficient photovoltaic cells used for solar power cost up to $50,000 per square meter. What if these cells could be replaced with a plastic solar concentrator less than 3 mm thick that concentrates sunlight 500 times at only $100 per square meter?
Diseases that cause blindness destroy the rods and cones in the retina. Ganglion cells rely on rods and cones to detect light as it comes into the eye. Could blindness be cured if ganglion cells could be coaxed by genetically engineered viruses to take on this function?
These tantalizing prospects are being pursued by two celebrated University of Rochester scientists whose work has already proven transformative, resulting in their election as 2021 fellows of the National Academy of Inventors.
All eyes on vision restoration with latest NEI Audacious Goals Initiative Grant
Juliette McGregor, Ph.D., assistant professor in the department of Ophthalmology, leads one of three new projects funded by the National Eye Institute's Audacious Goals Initiative (AGI) aimed at testing regenerative therapies for blindness due to retinal degeneration and monitoring transplanted cells as they integrate with host tissues.

Sara Patterson wins second place in the 2021 Steadman Family Postdoctoral Associate Prize in Interdisciplinary Research
Postdoctoral fellow Sara Patterson won second place in the Steadman Family Postdoctoral Associate Prize in Interdisciplinary Research. Sara competed against eight of the strongest postdoctoral fellow finalists at UR who delivered talks about their research. She will receive a $750 cash prize. Congratulations, Sara!
Dana Foundation Q&A with awardee Jesse Schallek
When you visit the ophthalmologist or optometrist to check your vision, the doctor will likely use an imaging tool called an ophthalmoscope to get a detailed look at the back of your eye, where the retina, optic nerve, and vasculature reside. The resulting orange-ish image, accentuated with meandering red veins and blood vessels, helps the doctor determine whether your eyes are healthy.
But it's possible such images could do even more. The retina is the only portion of the central nervous system (CNS) that can be observed from outside the body. Your eye doctor, in fact, can get a glimpse of it simply using a bright light and that ophthalmoscope. Jesse Schallek, Ph.D., has always been excited by the idea of finding a way to take images of living biology in its natural state. Today, using adaptive optics technology, a method first used to optimize long-range telescopes, he and his team have transformed the traditional ophthalmoscope so that it can now visualize the inner workings of the eye at the level of a single cell. The resulting technology offers a unique window into the CNS, which may help ophthalmologists understand the pathology of debilitating visual conditions like diabetic retinopathy and other forms of neurodegeneration and brain-related disease. Here, Schallek discusses how astronomy inspired his imaging advances, the challenges of imaging translucent cells, and how artificial intelligence can help give this kind of imaging approach even more clinical value.

Members of the Schallek Lab showcase their patented technologies at the Institute of Optics Industrial Associates Program
Video links:
Configurable aperture for biomedical imaging provides confocal and phase-contrast imaging
Single Cell Blood Velocity Software
2020

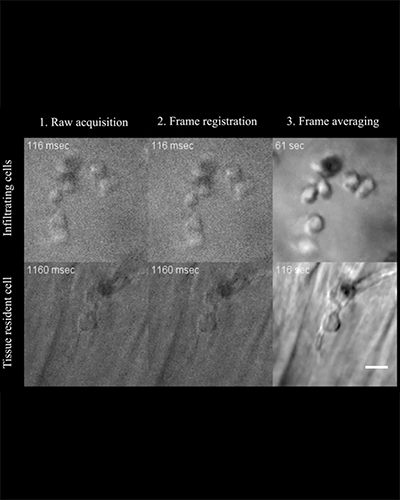
Imaging the secret lives of immune cells in the eye
Rochester researchers demonstrate way to track the interactions of microscopic immune cells in a living eye without dyes or damage, a first for imaging science.
Combining infrared videography and artificial intelligence, the new technique could be a 'game-changer' for some clinical diagnoses as well as for fields like pharmaceuticals.
Read more...
Kristen Bowles-Johnson received the NIH Loan Repayment Program Award
Congratulations to postdoc Kristen Bowles-Johnson, who received the NIH Loan Repayment Program Award worth $45,000.

David Williams selected as the recipient of the David F. Weeks Award for Outstanding Vision Research by the Association of University Professors of Ophthalmology (AUPO)
Congratulations to David Williams, who has been selected as the recipient of the prestigious David F. Weeks Award for Outstanding Vision Research by the Association of University Professors of Ophthalmology (AUPO) for 2021.
The RPB David F. Weeks Award for Outstanding Vision Research recognizes and celebrates an excellent vision researcher focused on potentially blinding eye disease. The "Weeks Award" has been established through the generosity of Research to Prevent Blindness (RPB), an anonymous donor, and the AUPO, to annually recognize and celebrate an outstanding ophthalmic vision scientist whose research has made meaningful contributions to the understanding and/or treatment of potentially blinding eye disease. The award carries the name of David F. Weeks, former President and Chairman of Research to Prevent Blindness, in honor of his contributions to the field of vision research. The award will be presented at the AUPO Annual Meeting in February 2021 and carries an unrestricted award of $50,000, payable to the recipient directly and to be used at his or her discretion.

Mina Chung, Retinal Surgeon and Researcher, Dies at 51
Mina Millicent Chung, M.D., an ophthalmologist and retina specialist who helped blind children to see and furthered the study of retinal disease, including macular degeneration, died Feb. 13 after a fall while skiing in Cortina d'Ampezzo, Dolomites, Italy. She was 51.
Chung was an associate professor of ophthalmology at URMC's Flaum Eye Institute and a faculty member in the University's Center for Visual Science. Before being recruited to Rochester in 2002, she completed a fellowship in vitreoretinal surgery at the University of Iowa College of Medicine, following residency and postdoctoral research at USC University Hospital in Los Angeles, where she served as chief resident from 2000 to 2002. Chung was a 1994 graduate of the Yale University School of Medicine who received her undergraduate degree from Yale in 1990.
Read more...
2019

Jesse Schallek featured in RPB Career Development Awards Video
ARIA faculty Jesse Schallek was featured in a video on the Research to Prevent Blindness Career Development Awards.
Read more...

David DiLoreto Jr. Selected to Chair University of Rochester Medical Center Department of Ophthalmology, Lead Flaum Eye Institute
David A. DiLoreto, Jr., M.D., Ph.D., was named chair of the University of Rochester Medical Center's Department of Ophthalmology and director of the Flaum Eye Institute, pending approval by the Office of the Provost. He succeeds Chair Steven Feldon, M.D., M.B.A., who will transition to associate vice president and director of the Office of Biomedical Research Development.
Read more...

Tyler Godat receives OSA FVM Young Investigator Award
Congratulations to Tyler Godat, who received the Optical Society of America Fall Vision Meeting Young Investigator Award for his presentation last Saturday in Washington DC. Tyler spoke about "In vivo classification of macaque foveal ganglion cells through optical recording of responses to chromatic and luminance flicker."

Jennifer Hunter elected OSA Fellow
Congratulations to Jennifer Hunter, who was elected a Fellow of the Optical Society of America.
Credit: Pixabay
ARIA on the Naked Scientist Podcast
Aby Joseph, Andres Guevara-Torres, and Jesse Schallek present on the Naked Scientist podcast (Start: 13:19 / End: 20:22). Read more...

Imaging That Twinkle in Your Eye: Assessing Vascular Health by Imaging Blood Cells in the Retina
Aby Joseph, Andres Guevara, and Jesse Schallek describe a new, noninvasive approach to assess vascular health in the journal eLife. Schallek's lab, part of the Flaum Eye Institute, developed a method to visualize how single blood cells flow through vessels of the eye using adaptive optics imaging.
Read more...

Congratulations to Dr. Sarah Walters, for her successful PhD defense
Congratulations to Dr. Sarah Walters, for her successful PhD Defense, "Two-Photon Excited Fluorescence Adaptive Optics Ophthalmoscopy of Retinal Function." Advised by Prof. Jennifer Hunter & Prof. David Williams
Abstract: The retina is the light-sensitive tissue at the back of the eye, which carries out the first steps in vision. Specialized neural cells in the retina known as photoreceptors are responsible for detection of light and its transduction by initiating an electrical signal to the brain. Adaptive optics scanning light ophthalmoscopy (AOSLO), which dynamically corrects aberrations of the ocular media in the living eye and affords a lateral resolution of 2 μm, has revolutionized our ability to visualize photoreceptors and many other microstructures in the retina. The implementation of two-photon excited fluorescence (TPEF) imaging in AOSLO has enabled not only complementary structural information throughout the retina, but an objective, non-invasive measure of visual function in photoreceptors by measuring TPEF kinetics from these cells.
The aim of the present thesis is to further develop and apply TPEF ophthalmoscopy as a novel measure of in vivo cellular function in the retina. First, TPEF ophthalmoscopy was used in conjunction with other imaging modalities to evaluate the extent of photoreceptor dysfunction in a non-human primate model of retinal degeneration. TPEF ophthalmoscopy was essential in determining that photoreceptors were non-functional. Second, the sensitivity of TPEF kinetics to detect changes in photoreceptor function in conditions relevant to disease pathogenesis was investigated. Systemic hypoxia was employed in non-human primates as a model of physiological change, reducing oxygen supply to the retina, and TPEF kinetics were shown to be slowed as a consequence. Finally, the capabilities of TPEF ophthalmoscopy were expanded by implementing intrinsic fluorescence lifetime imaging. TPEF lifetime imaging was shown to distinguish retinal cell classes that are functionally disparate, and lifetimes were altered in regions of retinal damage.
TPEF ophthalmoscopy has the potential to yield advances in understanding of both the basic physiology and pathology of the retina. If translated successfully into humans, TPEF ophthalmoscopy demonstrates promise as a valuable imaging modality that may, when used in conjunction with other clinical measures, identify early cellular dysfunction and longitudinally track pathological changes. Ultimately, it may assist in timely diagnosis, intervention, and development of treatments or vision restoration methods to combat blindness as a consequence of retinal disease.

Tyler Godat and Khang Huynh receive travel grants for ARVO
Congratulations to Tyler Godat and Khang Huynh, who received the ARVO Foundation/Retina Reseach Foundation/Joseph M. and Eula C. Lawrence Travel Grant to the 2019 ARVO Annual Meeting in Vancouver, British Columbia.
2018
- Aby Joseph and Sarah Walters received the ARVO Foundation/Retina Reseach Foundation/Joseph M. and Eula C. Lawrence Travel Grant to the 2018 ARVO Annual Meeting in Honolulu, HI
- Congratulations to Juliette McGregor, winner of the 2018 OSA FVM Young Investigator Award.
- July's Hajim Spotlight: Jennifer Hunter
- 'Bionic Eye' Helps Wyoming County Man See His Bride Again
- Congratulations to Aby Joseph, who won the best graduate student poster prize at the UR annual neuroscience retreat!
2017
- Congratulations to Aby Joseph, winner of the 2017 Leica Imaging Award for his time-lapse imaging of retinal microglia. Aby was honored with the award at the 2017 Immune Imaging Symposium at the University of Rochester.
- A 71-year-old woman blinded by an inherited disease recently thrilled University of Rochester doctors by reaching out and grabbing her ophthalmologist's hand after receiving a "bionic eye." The device allows her to distinguish light and motion, which she hasn't been able to do in decades. Read more...
- Congratulations to Ge Song, winner of the President's award for Engineering and Applied Sciences at the Undergraduate Research Exposition.
- In a study highlighted in the Proceedings of the National Academy of Sciences, Ethan A. Rossi, Ph.D., assistant professor of Ophthalmology at the University of Pittsburgh School of Medicine, describes a new method to non-invasively image the human retina, a layer of cells at the back of the eye that are essential for vision. The group, led by David Williams, Ph.D., Dean for Research in Arts, Sciences, and Engineering and the William G. Allyn Chair for Medical Optics at the University of Rochester, was able to distinguish individual retinal ganglion cells (RGCs), which bear most of the responsibility of relaying visual information to the brain. Read more...
2016
- Congratulations to Christina Schwarz, along with co-authors Robin Sharma, Brandi Hardy, David Williams, and Jennifer Hunter, for receiving tenth place in The Best Photo in Vision Research and Ophthalmology 2016.
- Jesse Schallek has been awarded the David Mahoney Neuroimaging grant from the Dana Foundation. The grant will support efforts to translate new metrics of blood flow evaluation into the clinical population.
- Congratulations to Juliette McGregor on receiving the Steadman Family Interdisciplinary Research Award. The award recognizes an outstanding Postdoctoral Associate or Postdoctoral Fellow at the University of Rochester whose research exemplifies the importance of cross-discipline approaches towards examining high impact questions of science.
- Congratulations to Andres Guevara for receiving the CONACYT fellowship award. The award supports his research that will develop new ways to extract optical contrast from highly translucent neurons of the retina.
- Guevara-Torres A, Williams DR, and Schallek JB (2015). Imaging translucent cell bodies in the living mouse retina without contrast agents. Biomed. Opt. Express 6, 2106-2119.
- Congratulations to James Feeks, recipient of an ARVO travel grant.
- David Williams has received the seventh annual Jay Pepose '75 Award in Vision Sciences from Brandeis University.
- Congratulations to Jesse Schallek, recipient of the Research to Prevent Blindness Career Development Award for his work on how blood flow, structures and cells inside the retina are affected by diabetes.
- Congratulations to David Williams, Gala Honoree at the ARVO Foundation & Dowling Society Gala Awards Ceremony and Dinner on April 30, 2016.
2015
- David Williams has been named the 2015 recipient of the Beckman-Argyros Award in Vision Research. Williams has also been awarded the Sigma Xi Procter Prize.
- Congratulations to David Williams, recipient of the 2015 Alcon Research Institute award for outstanding contributions in the field of vision research.
- Congratulations to Robin Sharma and Andres Guevara-Torres. Both received 2015 travel grants from ARVO.
- ARIA researchers David Williams, Jennifer Hunter, and Bill Merigan received a $3.8 million National Eye Institute grant for restoring vision through retinal regeneration.
- David Williams is a recipient of the 2015 ARVO Distinguished Service Award, which is presented to elected ARVO officers and editors-in-chief upon completion of their terms, in appreciation for dedicated service to ARVO.
- ARIA welcomes new principal investigator Jesse Schallek. Dr. Schallek is Assistant Professor of Ophthalmology, Neurobiology & Anatomy, and the Center for Visual Science.
2014
- A richly-deserved honor was bestowed on David R. Williams, Allyn Professor of Medical Optics and Director of the Center for Visual Science, who was elected to the National Academy of Sciences.
2013
- Robin Sharma's paper has been selected for Spotlight on Optics!
- Congratulations to Jesse Schallek for being selected as a Edmund Optics Grant Finalist!
- Congratulations to Dr. David Williams for winning the Edridge Green Medal from the Royal College of Ophthalmologists!
- ARIA has 3 postdoc opportunities available.
2012
- Dr. David Williams and his work on Adaptive Optics was featured in the Democrat and Chronicle [PDF]
- Congratulations to graduate student Robin Sharma for winning the the Emil Wolf Outstanding Student Paper Competition at FiO 2012
- Dr. David Williams and his work on Adaptive Optics was featured in the Rochester Review [PDF]
- Congratulations to Dr. David Williams for winning the 2012 António Champalimaud Vision Award, which recognizes novel approaches to imaging the eye
- Dr. Jennifer Hunter will be attending the 6th annual EOS Conference as an Invited Speaker
- Congratulations to Dr. Jesse Schallek, recipient of: Schmitt Program on Integrative Brain Research Postdoctoral Fellowship, 2012
Retina Research Foundation/J.M. and E.C. Lawrence Travel Award, 2012
ARVO Member in Training (MIT) Outstanding Poster Award, 2012